Biomarkers vs. Survival: Why Measuring Change Isn't the Same as Measuring Benefit
Biomarkers dominate headlines and health claims, but a change in a biomarker is not the same thing as a change in health. An examination of what biomarkers actually measure, when they reliably predict patient outcomes, and why promising results often fail to translate into real benefits.
Why biomarkers are everywhere
The rise of molecular biology and computational tools in medicine has led to an explosion in the discovery of molecular biomarkers involved in disease processes. Advances in genomics, proteomics, and data science have made it possible to measure thousands of biological signals simultaneously. Biomarkers offer a way to translate enormous datasets into something measurable and comparable.
Using biomarkers allows research to move faster and at lower cost. Instead of waiting months or years to observe a clinical outcome, researchers can quickly measure a short-term change in a biomarker associated with that outcome and infer a treatment’s potential effectiveness earlier. Regulators may accept biomarker changes as preliminary evidence, investors can interpret them as progress, and media coverage can frame them as breakthroughs. This has created an ecosystem where biomarker changes are often treated as synonymous with benefit.
But a change in a biomarker is not the same thing as a change in health.
What a biomarker actually is
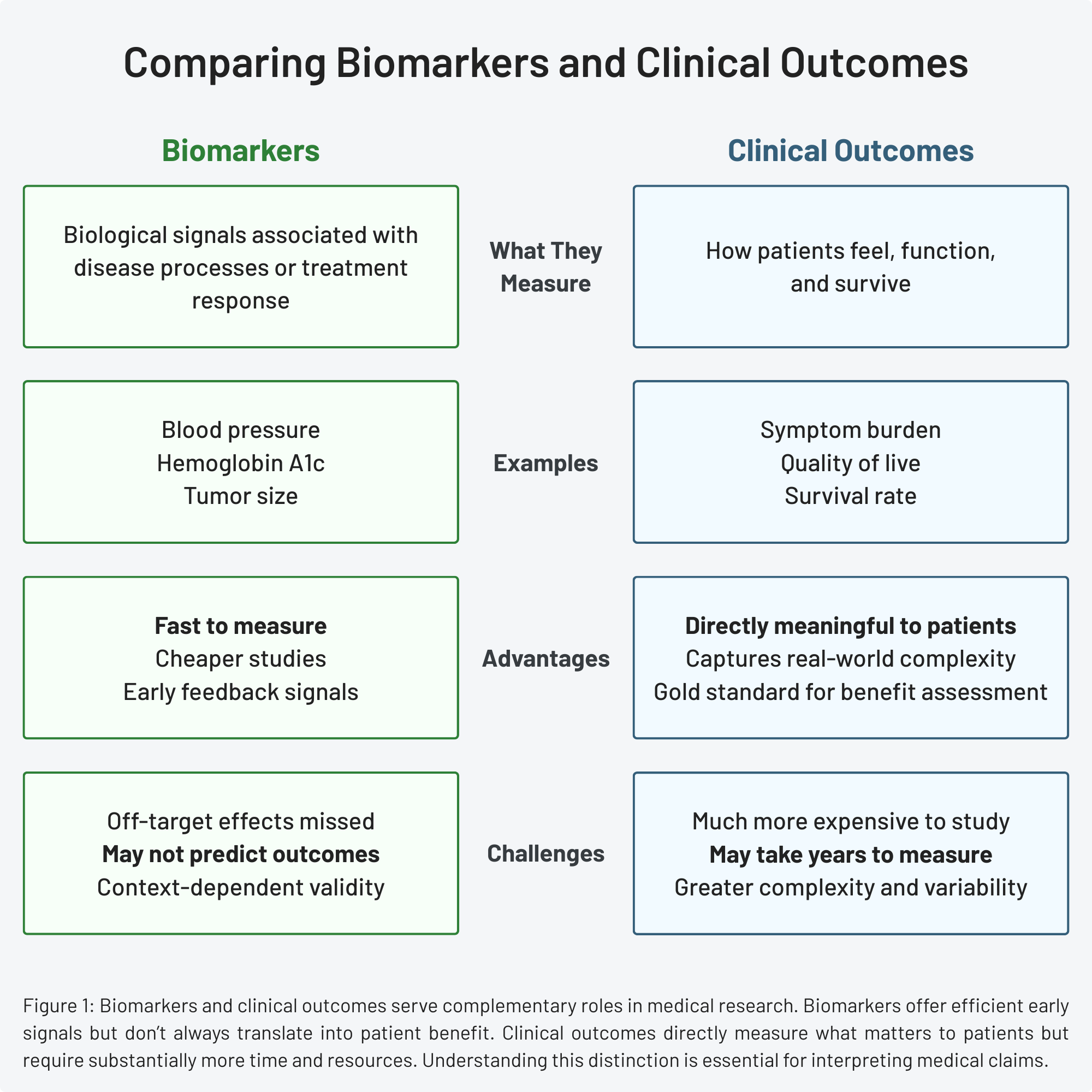
A biomarker, or biological marker, is an objective, quantifiable characteristic of a biological process. Common examples include cholesterol levels, blood pressure, hemoglobin A1c, and tumor size. These measures reflect aspects of underlying biological processes or disease states.
Biomarkers can be categorized by function: predictive, diagnostic, monitoring, response, prognostic, risk, and safety. Not all biomarkers are meant to be interpreted the same way. A diagnostic biomarker helps confirm the presence of a disease. A monitoring biomarker tracks changes over time. A prognostic biomarker estimates the likelihood of a future clinical event. Understanding these distinctions is important when evaluating research claims.
Biomarkers are among the most important tools in predictive medicine. They are usually faster and cheaper to measure than clinical outcomes and can provide valuable mechanistic insight. For this reason, biomarkers are often used as surrogate endpoints—or substitute markers, for how a patient will ultimately feel after treatment. Whereas long-term outcomes may take years to observe, surrogates can provide early feedback. If results are discouraging, a trial can be stopped early, potentially saving time and resources. This accelerates clinical research and drug development.
Clinical outcomes: what patients actually care about
Biomarkers are distinct from measures of how a person feels, functions, or survives—a category known as clinical outcome assessments. Clinical outcomes include symptom burden, quality of life, disease progression, functional capacity, and survival. These outcomes directly matter to a patient and are therefore considered the gold standard for evaluating treatment benefit.
Measuring clinical outcomes, however, is difficult. These studies require larger sample sizes, longer follow-up periods, and substantially more resources. A biomarker study might enroll 200 patients and conclude within six months. An outcome study might require 10,000 patients followed for five years or more. The difference in cost, complexity, and risk is substantial. Researchers typically need strong preliminary evidence before launching these studies.
Clinical outcomes are also inherently messier. Survival is influenced by many interacting factors. Quality of life depends partly on subjective experience. These measures reflect how an intervention performs in the complexity of real life, not just under controlled experimental conditions. While they’re more difficult to study, they’re far more meaningful to patients.
If you want a faster way to read health studies without oversimplifying them, subscribers get a free guide: How to Read a Health Study in 10 Minutes.
When biomarkers do predict outcomes
Not all biomarkers function as surrogate endpoints. Only a small subset have been validated to predict clinical outcomes reliably. For a biomarker to serve as a valid surrogate, several conditions must be met.
First, changes in the biomarker must consistently correlate with changes in the clinical outcome across multiple studies and populations. Second, the biomarker must lie on the causal pathway of the disease, not just track alongside it. Correlation alone is insufficient—changing the biomarker should help explain changes in disease risk. Third, different interventions that improve the biomarker through distinct mechanisms should also improve the clinical outcome. If a biomarker improves under one treatment but not another, its value as a surrogate is limited to specific contexts.
Blood pressure represents a well-validated surrogate. Decades of research have established that lowering blood pressure reliably reduces the risk of heart attack and stroke. This is consistent across different pharmacologic interventions—regardless of which mechanism a drug uses to lower blood pressure, the benefit remains. The biomarker sits directly on the causal pathway: elevated blood pressure damages blood vessels over time, so reducing pressure prevents damage. We can be highly confident that the effect on blood pressure, and not another property of the drugs, mediates the benefits seen in this context.
Even when validation criteria are met, surrogacy only applies to specific scenarios. A biomarker may predict outcomes for a particular disease stage or drug class but fail in others. Surrogacy is not a permanent designation but rather a context-specific judgment that requires ongoing validation. This specificity matters when new treatments emerge.
When biomarkers mislead
There are several reasons a biomarker may fail to translate into meaningful health benefits. The biomarker may be downstream of the disease process rather than causal. An intervention may change the marker but not the underlying disease. The disease may be more complex than any single biomarker can capture. Or off-target effects may negate potential benefits.
Tumor shrinkage provides a classic example. It is commonly used to assess the effectiveness of cancer treatments. Intuitively, reducing tumor size seems like it should improve survival—smaller tumors should take longer to reach life—threatening thresholds. But this logic ignores factors such as cell proliferation rates, genetic mutations, and aggressive tumor recurrence. Many therapies successfully shrink tumors without improving long-term survival. In some cases, treatments may even trigger more aggressive regrowth, a phenomenon researchers are still working to understand.
This pattern is not unique to oncology. Improvements in laboratory markers do not always translate into reductions in mortality. Hemoglobin A1c (HbA1c), a widely used marker of glycemic control in diabetes, illustrates this limitation. Although better glycemic control is generally assumed to lower mortality risk, an analysis of more than 6,000 adults with diabetes found that despite improved HbA1c control rates between 1999 and 2014, cardiovascular and all-cause mortality rates did not decline over the same period.
This finding does not mean that glycemic control is irrelevant. It means that diabetes is more than blood sugar alone. Cardiovascular health in diabetes depends on blood pressure, lipid levels, inflammation, kidney function, and numerous other factors. Focusing on a single biochemical marker cannot fully capture the complexity of disease risk or treatment benefit.
Why biomarkers dominate headlines anyway
Given these limitations, why do biomarker-based studies receive so much attention? The answer involves both human psychology and research economics.
The human body is complex, and numbers are reassuring. A biomarker that changes predictably feels more concrete than overall health, which can be highly variable and difficult to quantify. Biomarkers are easier to explain, easier to celebrate, and easier to frame as early wins. When cholesterol drops by 100 points, when blood sugar normalizes, when a tumor shrinks by 50%—these feel like victories, and sometimes they are. Often, they are only part of the story.
From a research perspective, biomarker studies offer practical advantages. Pharmaceutical companies can obtain preliminary signals about drug efficacy in months rather than years. Academic researchers can publish findings and advance their careers without waiting a decade for outcome data. Regulatory agencies can grant accelerated approval based on biomarker improvements, with the understanding that confirmatory outcome trials will follow. In environments that reward clarity and speed, biomarkers become an appealing shorthand, even when they can overstate the strength of the evidence.
How to read biomarker-based claims more carefully
There are a few key questions to ask when interpreting biomarker-based claims:
- What did the study actually measure: a biomarker or a clinical outcome?
- Is the biomarker validated for predicting that outcome?
- How long was the follow-up?
- Is this early-phase or late-phase evidence?
Paying attention to both statistical significance and real-world relevance can help calibrate expectations.
Why this matters
Biomarkers are powerful tools, but they are intermediate measures, not the finish line. When confidence is misplaced in biomarkers, resources may be devoted to interventions that ultimately fail to improve real outcomes. Patients may develop expectations that later evidence can’t support. Over time, this cycle can create the impression that science is inconsistent or unreliable, when in reality, the evidence was never as strong as it appeared.
The strongest conclusions emerge when biomarker data are combined with meaningful clinical outcomes. Understanding what biomarkers can—and can’t—tell us helps us evaluate health claims more accurately, ask better questions about medical research, and engage with science more honestly.
References
- Predicting Clinical Outcomes Using Molecular Biomarkers, Biomarkers in Cancer
- What are Biomarkers?, Current Opinion in HIV and AIDS
- Biomarker definitions and their applications, Experimental Biology and Medicine
- Blood pressure as an example of a biomarker that functions as a surrogate, AAPS Journal
- Paradoxical Effects of Tumor Shrinkage on Long-Term Survival of Cancer Patients, Frontiers in Applied Mathematics and Statistics
- Diabetes management dilemma: association between glycated hemoglobin levels and mortality risk in diabetic patients, Cardiovascular Diagnosis & Therapy

