Why Most Mouse Studies Don’t Become Medicine
Mouse research has powered major medical advances. It has also led to many promising ideas that never worked in humans. A closer look at when mouse studies are useful and when headlines get ahead of evidence.
Why mouse studies dominate early research
Mice are the most commonly used animals in biomedical research. They’re used to study diseases, behaviors, nutrition, aging, and genetics, among many other areas. Beyond being genetically similar to humans, they’re small, inexpensive to house, have short reproductive cycles and lifespans, and are generally mild-tempered. These traits make them practical and scalable research tools, especially in early-stage experiments where many iterations are needed.
Researchers can gain significantly more information about biological pathways and disease mechanisms by studying a complex living organism than by relying solely on computer models or cell cultures. Living systems capture interactions between tissues, immune responses, metabolism, and environmental exposures in ways that simplified models cannot. For many questions, animal models represent an important step between isolated cells and real-world human disease.
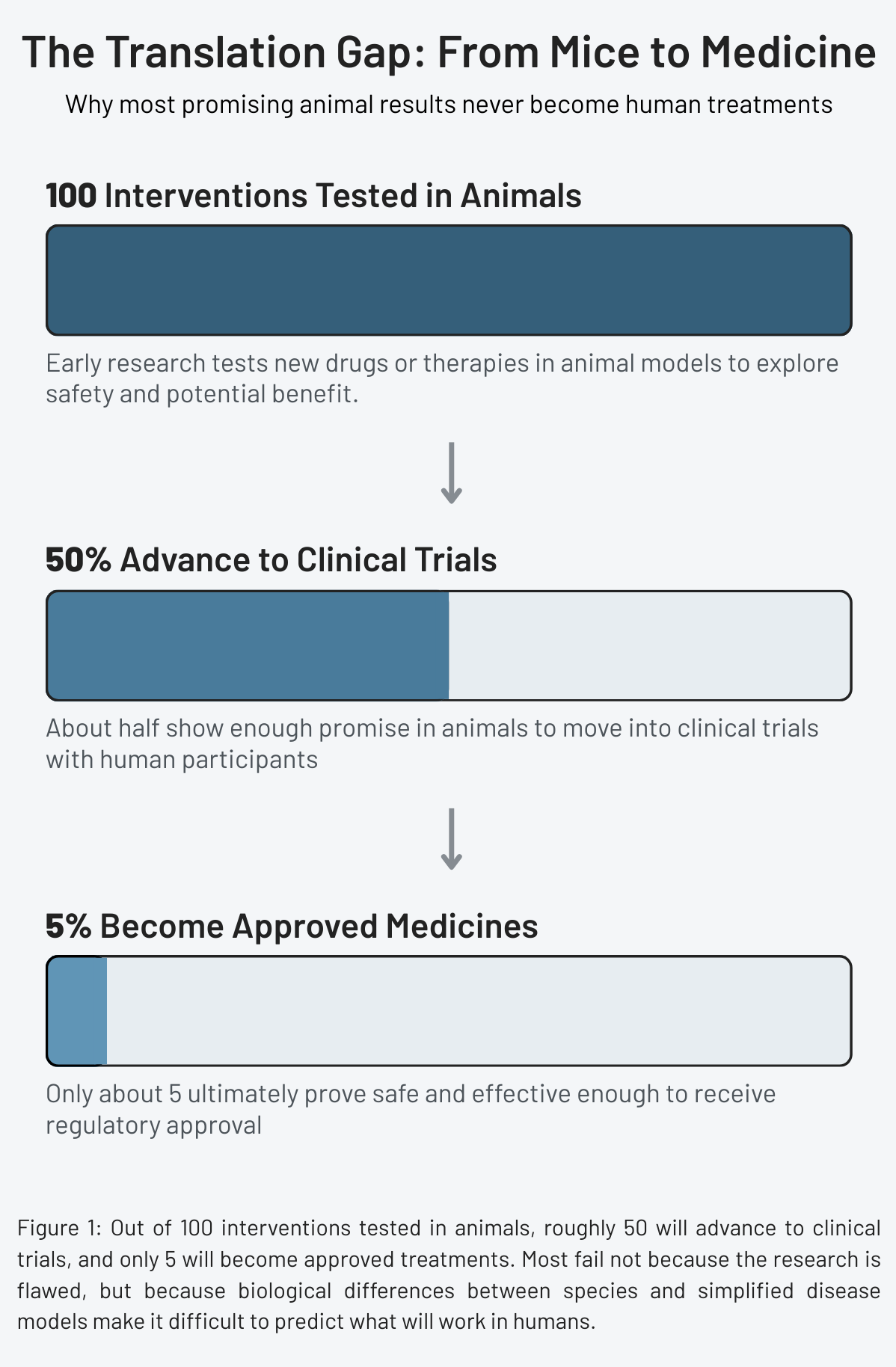
While mouse studies have contributed to many important advances, a large body of evidence suggests their predictive power is limited. A widely cited review found that about 50% of therapies progress from animal studies to human trials, but only 5% ultimately reach regulatory approval. In other words, most interventions that look promising in animals fail somewhere along the path to clinical use. Given this low translational rate, why are mouse models still so heavily relied on?
What mouse studies are actually designed to answer
To understand this gap, it helps to clarify what mouse studies are (and are not) designed to do.
Mouse studies are primarily used to investigate biological mechanisms, test gene function, and evaluate the basic safety and potential efficacy of new interventions before clinical trials. They function as proof-of-concept experiments, helping researchers determine whether a therapeutic approach is biologically plausible and worth pursuing further.
These studies allow scientists to manipulate variables in ways that would be impossible or unethical in humans. Researchers can knock out genes, induce disease states, and examine tissues at time points that would never be feasible in living people. This level of control makes mouse models powerful tools for answering focused mechanistic questions.
Because mouse studies are highly controlled, it's easier to identify cause-and-effect relationships. Researchers can isolate the role of a specific gene, signaling pathway, or exposure with minimal interference from confounding variables. However, this strength is also a limitation. Human disease rarely unfolds in controlled conditions. It reflects a complex interplay of genetics, environment, behavior, and chance over time.
As a result, conclusions drawn in mice should not be immediately extrapolated to humans. Mouse studies are best viewed as hypothesis-generating rather than hypothesis-confirming. They can suggest what might work, but they cannot reliably predict what will work in people.
If you want a faster way to read health studies without oversimplifying them, subscribers get a free guide: How to Read a Health Study in 10 Minutes.
The evolutionary distance problem
One of the most fundamental challenges in translating mouse research is evolutionary distance. Human and mouse lineages diverged from a common ancestor approximately 65-75 million years ago. Over that time, evolution has preserved many core biological processes while also introducing important differences.
Although about 90% of human and mouse genomes reside in conserved regions, only around 40% of nucleotides align exactly. Humans and mice genomes each have roughly 20,000-22,000 protein-encoding genes, with a high degree of gene orthology: about 80% of human and 72% of mouse protein-coding genes have a one-to-one orthologous counterpart. This means many genes are related and often functionally similar–but not identical.
Crucially, similarity exists on a spectrum. Some genes are 99% identical across species, while others share closer to 60% similarity. Even when genes are conserved, their expression patterns, regulatory controls, and interactions with other genes can vary. Small differences at the molecular level can cascade into large differences in physiology or disease behavior.
Beyond genes and transcripts, differences in cell types and organ complexity also complicate translation. For decades, it was believed that humans had around 200 distinct cell types. Advances in single-cell RNA sequencing have now identified more than 400, with hundreds more likely to be discovered. Differences in cell composition and relative abundance within organs can introduce meaningful variation between mice and humans. Importantly, these differences can be masked when researchers analyze whole tissues rather than cell-specific responses.
Physiological differences further complicate translation. Mice have faster metabolisms, distinct immune system components, and different hormonal regulation. Their smaller size affects how drugs are absorbed, distributed, and eliminated. A mouse heart beats around 500-600 times per minute, compared to a human’s 60-100 beats per minute. These differences can profoundly influence disease progression and treatment effects.
Humans and mice share many genetic similarities, but depending on the specific biological process being studied, mice may differ in ways that limit their usefulness as models.
The laboratory mouse isn’t a 'normal' mouse
Several types of laboratory mouse models are used in research. Transgenic mice carry genes inserted from another species. Knock-in and knock-out mice are engineered so specific genes are activated or disabled. Inbred mice are produced through many generations to be genetically identical.
These models make it easier to isolate the effects of individual genes or pathways, but they also lack the complexity of many human diseases. Genetic variation among humans is high—up to about 0.5%—and can meaningfully affect gene expression, disease risk, and treatment response. This diversity is difficult to model in laboratory mice.
Most lab mice are also housed in highly controlled, pathogen-free environments that don’t resemble human microbial exposures. This artificial setting can alter immune development and function in ways that don't reflect real-world conditions.
Inbred strains introduce another limitation: they’re essentially clones. Uniform genetic backgrounds reduce experimental variability, but they also produce uniform responses to treatment–responses that may not generalize to a genetically diverse human population.
Disease models are simplified versions of complex human conditions
Two critical features determine how useful an animal model is: how accurately it reflects a disease’s cause (etiology) and how closely it mirrors the clinical presentation.
Asthma provides a useful example. Asthma is a chronic condition characterized by episodes of wheezing and breathlessness, with symptoms and severity varying widely between individuals. It’s increasingly recognized not as a single disease, but as a heterogeneous syndrome comprising multiple phenotypes.
Mice cannot naturally develop asthma. Instead, researchers induce allergic airway inflammation to approximate certain aspects of the condition. While these models can be informative for studying specific immune pathways, they fall short in important ways. Mice have different airway cell compositions, lack the extensive pulmonary circulation seen in humans, and show strain-specific immune responses to allergens.
Most importantly, mouse models cannot capture the full range of environmental exposures, genetic susceptibility, microbial influences, and long-term disease progression that shape asthma in humans. As a result, many treatments that appear effective in mouse models of asthma have failed to deliver meaningful benefits in clinical trials.
This pattern repeats across many complex, multifactorial diseases, including neurodegenerative disorders, metabolic diseases, and psychiatric conditions.
Methodological issues in mouse research
Beyond biological differences, methodological issues further limit translation.
Many animal experiments suffer from design and reporting limitations. Studies may be underpowered or insufficiently blinded, increasing the risk of bias. Ethical and regulatory constraints often limit sample sizes, which can reduce statistical power and increase the likelihood of false-positive findings.
Animal studies are also less frequently synthesized through systematic reviews and meta-analyses than human studies. This is a significant problem, as animal research often provides the justification for human trials. Without rigorous synthesis of preclinical evidence, researchers may advance hypotheses based on misleading or incomplete data.
Publication bias affects animal research just as it does human studies. Positive findings are more likely to be published than negative results, creating a distorted literature where successful mouse experiments are overrepresented. This can make interventions appear more promising than they truly are, encouraging costly human trials with low chances of success.
When mouse studies do translate
Despite these limitations, mouse research has enabled important medical advances. From cancer immunotherapies to treatments for rare genetic disorders, many successful interventions originated in mouse models. The key is understanding when mouse models are most likely to be predictive.
Mouse studies tend to translate best when they focus on highly conserved biological mechanisms. Fundamental processes such as DNA repair, basic immune signaling, and core metabolic pathways often behave similarly across species. In contrast, studies of complex behaviors, age-related diseases, and conditions involving many interacting factors are less likely to translate directly.
Single-gene disorders show particularly high translation rates. When a disease arises from a specific mutation, a mouse engineered with that same mutation often reproduces key features of the human condition. Cystic fibrosis research is a well-known example: mouse models helped researchers understand disease mechanisms and evaluate therapies that ultimately benefited patients.
Drug safety testing in mice, while imperfect, has also prevented many toxic compounds from advancing to human trials. Although mice don’t predict all human toxicities, they identify enough risks to remain a critical early filter.
How to read mouse study headlines more carefully
When you encounter headlines touting results from mouse studies, ask a few follow-up questions:
- What type of mouse was used?
- What was studied—a gene, a pathway, or a complex disease process?
- How strong are the biological parallels between mice and humans in this context?
- Has the finding been independently replicated?
Why this matters
Mouse models remain foundational tools in basic and preclinical research. Problems arise not from their use, but from overinterpretation. A positive result from a mouse study is interesting—and sometimes exciting—but it should be viewed as one piece of evidence, not a conclusion.
This has implications for science funding and research priorities. If large investments in mouse research yield limited translation, it’s reasonable to ask whether resources should be more evenly distributed across human-relevant approaches, such as advanced cell systems, organ-on-a-chip technologies, and carefully designed human studies.
For patients and advocates following research that affects them or their loved ones, understanding these limitations helps calibrate expectations. Promising mouse results deserve attention, but not certainty. The path from mouse findings to human therapy is long, uncertain, and failure-prone. Recognizing this doesn’t diminish the value of mouse research—it places it in context as one step in a complex process of discovery.
References
- Analysis of animal-to-human translation shows that only 5% of animal-tested therapeutic interventions obtain regulatory approval for human applications, PLoS Biology
- Why animal studies are often poor predictors of human reactions to exposure, Journal of the Royal Society of Medicine
- The (misleading) role of animal models in drug development, Frontiers in Drug Discovery
- Commonly Used Animal Models, Principles of Animal Research for Graduate and Undergraduate Students
- Comparative transcriptomics in human and mouse, Nature Reviews Genetics
- The Applicability of Mouse Models to the Study of Human Disease, Methods in Molecular Biology

